A 71 year old male with a history of COPD and heavy ongoing smoking habit, presented to the emergency department with a 3 hour history of tongue, mouth and throat swelling. He had awoken from an afternoon nap with the symptoms and initially refused to come to hospital. By the time he arrived he was unable to contain his tongue in his mouth, he had dramatic submandibular swelling and stated he was unable to swallow. There were no urticarial symptoms or wheeze. The swelling was not warm or tender.
Medication history was notable for including an ACE inhibitor and lots of inhalers. He denied any allergies in the past.
The most likely diagnosis was ACE I angiooedema, but the possibility of allergy, tongue base tumour and acquired angiooedema were entertained. A trial of nebulized adrenaline, IM adrenaline and IV steroids were initiated with little hope of success while we prepped for a challenging airway. We were lucky enough to have a very experienced anaesthetist with an interest in and years of experience in, managing difficult airways.
After 30 minutes of prepping and briefing whilst hoping that the trail of adrenaline may help it was decided that the symptoms were worsening despite our best efforts and that his airway should be managed and the drug allowed to wear off.He underwent awake nasal fibreoptic intubation and the views the scope revealed were of a very swollen oropharynx, his cords remained normal in appearance.
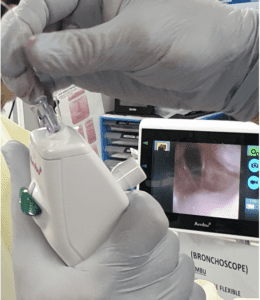
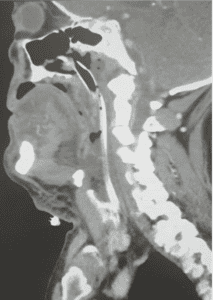
He remained intubated for 4 days until the oedema settled and he was uneventfully extubated.
Discussion
There is a lot to discuss in this case.
The techniques of awake nasal intubation is complex.
Icatibant was suggested as a possible treatment for ACE I mediated angiooedema due to its bradykinin inhibiting role, but the clinical experience has not fulfilled the early promise. A double blind trial by Bas et al (5) demonstrated a more rapid resolution of symptoms in the icatibant treatment group, but was not powered to look at intubation rates. This was an industry and government funded, multi-centre, double-blind, double-dummy randomized phase 2 study. Patients in the treatment group received icatibant 30 mg subcutaneously and those in the control group (standard therapy group) received prednisolone 500 mg ( a MASSIVE dose) and clemastine 2 mg (an antihistamine) intravenously. Patients assessed the intensity of symptoms at several intervals within 48 hours. Blinded investigators further assessed the signs and symptoms. If there was no symptom improvement at six hours after treatment administration, a rescue therapy of icatibant and prednisolone was administered irrespective of the study group. A final follow-up was performed at day 14 after discharge.
But a subsequent similar trial did not demonstrate any benefit. (6)
Given the excessive cost, lack of availability and usual provision as a named patient only medication under PBS it is probably only worth using icatibant on those “kitchen sink” patient who you are throwing all possible treatments at.
The indications for icatibant include
Patient must have confirmed diagnosis of C1-esterase inhibitor deficiency,
AND
Patient must have been assessed to be at significant risk of an acute attack of hereditary angioedema,
AND
The condition must be assessed by a clinical immunologist; OR
The condition must be assessed by a respiratory physician; OR
The condition must be assessed by a specialist allergist; OR
The condition must be assessed by a general physician experienced in the management of patients with hereditary angioedema.
None of these allow use in ED for non hereditary angiooedema and having been through the process of trying to get it out of hours for acquired C1 esterase inhibitor deficiency I can say it is often easier to manage the difficult airway than PBS authority. It is obviously not in the patients interest to resort to emergency cricothyroidotomy rather than a simple injection, but that is the system in which we are obliged to practice.
The heart of the treatment of ACE I medicated angiooedema is the early recognition and competent airway management whilst being aware that is it likely to be tiger country in there!
ACE Inhibitors
ACE-inhibitor-related angiooedema is not a true allergy but is considered an adverse class effect secondary to the mechanism of action (1) The proposed pathophysiologic mechanism of ACE I medicated angiooedema is the accumulation of bradykinin secondary to the inhibition of ACE. Questions persist as to why ACE-inhibitor-mediated angiooedema can occur anytime from hours to years after a patient has started the medication.
Angiooedema due to ACE inhibitors commonly occurs within the first few weeks of treatment but has been reported up to 7 years after the start of this medication. In 1999, Gibbs et al described 70% of angioedema cases occurring within 4 weeks of starting ACE inhibitor treatment, as well as found three cases where angioedema occurred greater than 24 months after the start of an ACE inhibitor (2).This may be explained by the fact that in the setting of ACE inhibition, aminopeptidase P (APP) and dipeptidyl peptidase (DPP4) become responsible for breaking down the vasoactive peptides bradykinin and substance P, respectively.
The proposed mechanism by which ACE interact with the vasoactive substances is shown in the (complex) diagram below.
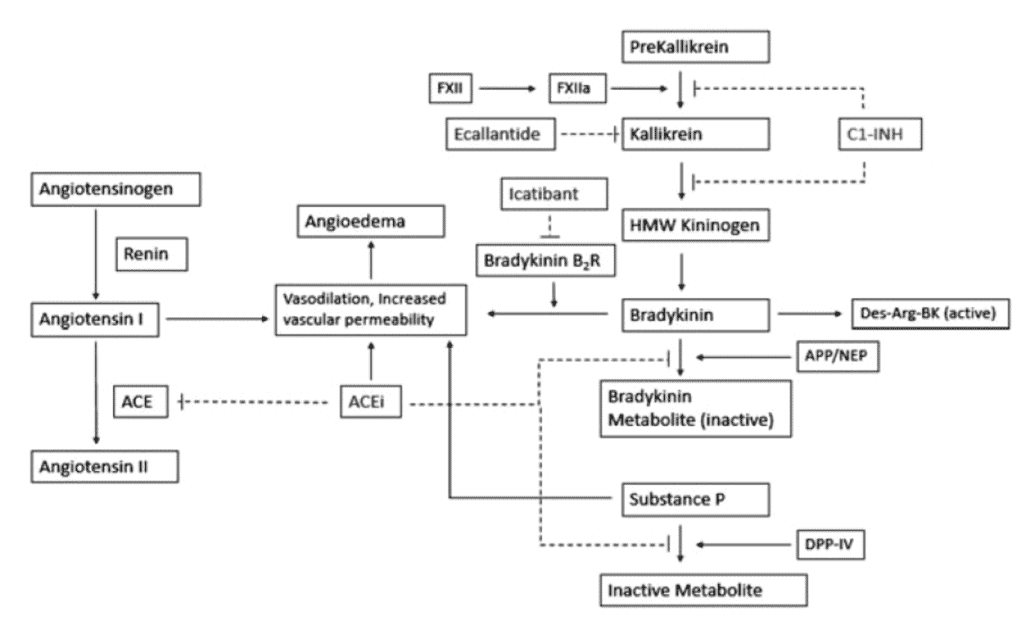
Thus in the presence of ACE I there is an increased half-life of bradykinin (usual half-life 17secs), increased levels of angiotensin I and substance P all of which increase the chances of angiooedema from a trivial trigger.
Factors adversely affecting these enzymes may trigger angioedema. Proposed factors include female gender, African descent, and tobacco use (4). Certain atopic diseases, such as seasonal allergies and asthma, may also be risk factors. On the other hand, diabetes mellitus is possibly associated with a decreased risk because diabetic patients appear to have increased DPP4 activity to aid in the breakdown of substance.
References
- Winters et al.. Emergency department management of patients with ACE-inhibitor angioedema. I Emerg Med 2013;45(5):775-80
- Gibbs et al. Angioedema due to ACE inhibitors: increased risk in patients of African origin. BrJ Clin Pharmacol 1999; 48: 861-5.
- Hirschy et al. Treatment of Life-Threatening ACE-Inhibitor-Induced Angioedema. Advanced Emergency Nursing Journal. Volume 40(4), October/December 2018, p 267-277
- Kostis et al. Incidence and characteristics of angioedema associated with enalapril. Arch Intern Med 2005;165(14); 1637-42.
- Bas M et al. A randomized trial of icatibant in ACE-inhibitor-induced angioedema. The New England Journal of Medicine, 372(5), 418-425.
- Sinert et al. Randomized trial of icatibant for angiotensin-converting enzyme inhibitor-induced upper airway angioedema. Journal of Allergy and Clinical Immunology: In Practice, 5(5), 1402-1409
Guest Blog By Dr Will Davies