A 50 year old female presented to the emergency department with anxiety, palpitations and feeling generally unwell. She had no past medical history and was on no medications. She had, however, seen her GP the week before complaining of 2 weeks of palpitations, anxiety and weight loss. Her GP had checked her thyroid function tests and started her on carbimazole 3 days previously. However, since then her symptoms had worsened. In particular, she was having palpitations, sweats, anxiety, and diarrhoea.
On examination, she looked anxious and jittery with a heart rate of 115, a respiratory rate of 22, and other vital signs which were normal. She was afebrile. She had a fine tremor and the examination was otherwise unremarkable. There was no signs of goitre, proptosis or oedema. There was no clinical evidence of heart failure and her ECG showed sinus tachycardia.
Review of bloods from the GP showed:
TSH <0.01 (0.3-4.5 mU/L)
T3 9.6 (3.0-7.2 pmol/L)
T4 15 (7-17 pool/L)
She had also had an ultrasound of her neck, that showed a nodule in her thyroid.
So, it is clear that the patient is thyrotoxic. The questions this raised in my mind were
- Why is she getting worse in spite of carbimazole?
- Does she need a radioiodine scan?
- Where is the line between thyrotoxicosis and thyroid storm?
To understand the answers to these questions, we first need to understand the physiology.
Thyroid hormone synthesis is comprised of a number of steps, all of which are TSH dependent
- Iodine taken up by the thyroid
- Iodine is oxidised and organified
- The iodine is then bound to thyroglobulin.
The last 2 steps are controlled by thyroid peroxidase
Next, Thyroid Hormone (TH) is released, and circulating TH is 99% bound to thyroxine binding globulin
There are 2 forms of TH, T3 and T4. T3 is more potent than T4, and about 10-20% of circulating T3 is released from the thyroid, the rest is converted from T4 by peripheral deiodonases. (If you are a member, there is more on Physiology at primaryexamcourse.com )
We need to understand the physiology to understand the different forms of thyrotoxicosis and thus the role of antithyroid drugs
There are 2 basic causes of thyrotoxicosis
- Diseases of increased TH synthesis (Grave’s disease, Toxic adenomas and toxic multinodular goitre)
- Diseases of increased release of preformed TH (viral thyroiditis, Hasimoto’s, which can have an initial hyperthyroid phase due to inflammation releasing TH, DeQuervains’ thyroiditis etc).
This is important to understand because the antithyroid drugs (carbimazole and PTU) work to decrease TH synthesis, so they work for the first group of diseases and NOT for the second group. PTU does decrease peripheral conversion of T4 to T3 but this is a minor effect.
So, how do we work out the cause of this patient’s thyrotoxicosis?
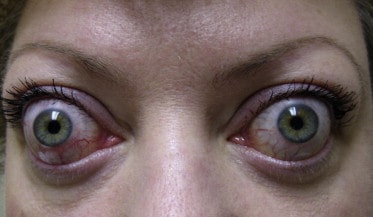
Firstly, we may see clinical features of Grave’s (rapid onset, exophthalmos, diplopia, pretibial myxoedema, acropachy (soft tissue swelling of hands and clubbing of fingers)).
Next, we correlate with bloods. Patients with thyrotoxicosis typically have raised T4 and supressed TSH (though some can have isolated high T3 and normal T4, so called ‘T3 thyrotoxocosis’, and rarely the TSH will be high if there is a TSH secreting pituitary adenoma)
Then we look at antibodies. TSH-Receptor antibodies (TRAB) are almost universal in Grave’s disease, but can be present in Hashimotos. Anti-TPO antibodies and thyroglobulin antibodies can be present in Grave’s disease and Hashimotos so are poorly discriminatory – see the below table.

Finally, if the cause is not clear from clinical features and bloods, we need a radionuclide scan. Grave’s will show diffuse uptake (A, below), nodules including MNG will show up as isolated hot spot/s (B and C), and Hashimotos will show up as decreased uptake (D).
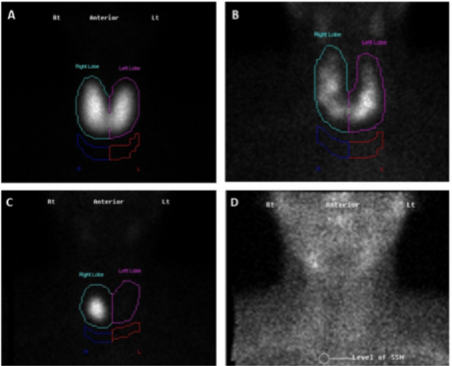
Fig 3, Vaidya, B and Pearce, S, “Diagnosis and Management of Thyrotoxicosis, BMJ 2014;349:g5128
Next, how do we treat thyrotoxic patients?
Antithyroid drugs are useful for conditions of increased TH synthesis (Graves disease, toxic adenomas and toxic multinodular goitre) but not for other causes of thyrotoxicosis. These diseases are managed by beta blockers and symptomatic treatment (see below). However, given that it often takes time to define the cause, it is reasonable to start antithyroid drugs early. Carbimazole is the drug of choice due to better compliance (once a day dosing), and the risk of fulminant hepatitis with PTU (about 1 in 10,000). Carbimazole is however teratogenic, and has a risk of agranulocytosis, so ask patients to present if they get symptoms of agranulocytosis (fever, sore throat and mouth ulcers). PTU is the drug of choice for patients who may become pregnant, and also in thyroid storm (as it decreases peripheral conversion of T4 to T3).
Antithyroid drugs take 5-10 days to have any effect, so consider starting other treatments in the interim. β-blockers are useful for palpitations, tremor and anxiety and propranolol is the drug of choice here. Benzodiazepines are occasionally needed for the anxiety/agitation.
Finally, what about thyroid storm?
This is an exaggerated form of thyrotoxicosis with a high (10-30%) mortality. Treatment needs to be started urgently on clinical grounds, without relying on confirmatory investigations. However, there are no universally accepted clinical criteria and what’s more, the T3, T4 levels are not always higher in thyroid storm when compared to uncomplicated thyrotoxicosis!
So, how do we diagnose storm? There are some clinical features to look out for
- Fever is almost universal
- Tachycardia is out of proportion to the rest of the clinical signs
- Look for complicating features too
- AF, MI, heart failure, shock
- Agitation delirium coma.
There is a scoring system from Burch and Wartofsky which might help, but this isn’t universally accepted either.
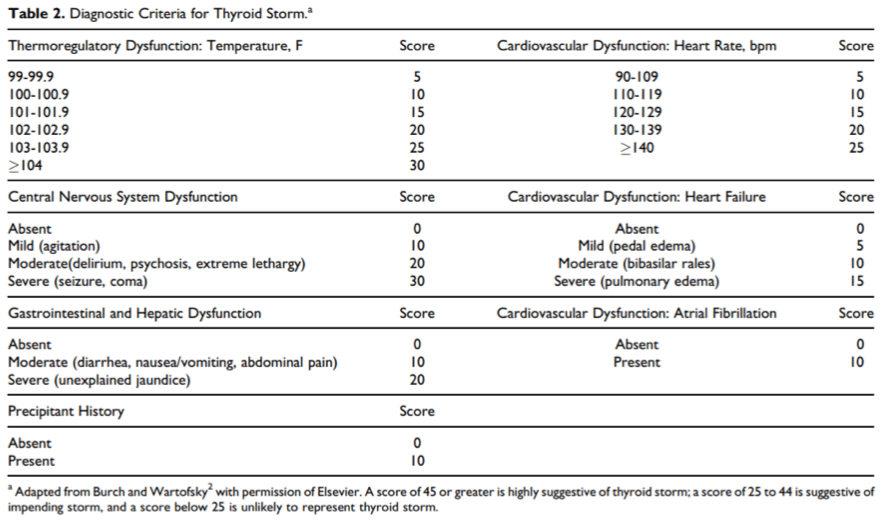
From Chiha M., et al, “Thyroid Storm: An Updated Review”, J Intensive Care Med 2015, Vol 30(3) 131-40
Treatment of Thyroid Storm involves
- Stop new TH production
- high dose antithyroid medications, PTU preferred
- Inhibit peripheral effects of TH
- Propanolol is the β-blocker of choice, and also decreases conversion of T4 to T3
- Inhibit TH release: Lugol’s iodine (must be given >30 mins after PTU to avoid it being substrate for the storm)
- Stress dose glucocorticoids (the HPA axis is relatively depressed)
- As well as supportive care (fluids, cooling etc)
So, what happened to our patient?
We treated her with propranolol with a good response, and she was discharged that afternoon. She went on to have a radionuclide scan which showed a hot nodule, so she has had radioiodine treatment. She remains on her carbimazole and propranolol with excellent symptom control.
Adam will be speaking on Thyroid Disorders at EMCORE Gold Coast.

Dr Adam Michael DEMT Bundaberg Hospital
References
- Vaidya, B and Pearce, S, “Diagnosis and Management of Thyrotoxicosis, BMJ 2014;349:g5128
- Chiha M., et al, “Thyroid Storm: An Updated Review”, J Intensive Care Med 2015, Vol 30(3) 131-40
- Franklyn, JA and Boelaert, K, “Thyrotoxicosis”, Lancet 2012 Vol 379(9821): 1155 – 1166.







Excellent article, thank you. Can I throw in a couple of pointers from the only storm I recall seeing, peripheral hospital a few years back. 30 year old lady presented with nausea and vomiting. That was it. Incidentally, she was being monitored for thyroid disease, s/b professor of endocrinology a month ago, all tests were normal. She had gained at least 20kg, BMI easily over 30. HR 150 at rest with a tremor.
I looked it up. Up to a third of thyroid storms present with weight GAIN because the hyperphagia can trump increased BMR.
Next investigation – I couldn’t believe that she’d go from euthyroid to storm in a month. Rang every laboratory and couldn’t find a recent test. Finally phoned her endocrinologist and asked him and he checked and was chagrined to realise that he had looked at results that were 6 months old.
Clerical errors can be lethal, and we all make them.
It’s a nice article. I cleared out all my doubts about thyrotoxicosis and got a complete idea of thyrotoxicosis. Thanks for sharing.
Thyrotoxicosis – Resus
abmzeckfynx
bmzeckfynx http://www.g1d8l662xq0x87jd7bw8l99ee0163exjs.org/
[url=http://www.g1d8l662xq0x87jd7bw8l99ee0163exjs.org/]ubmzeckfynx[/url]
最新ブランドスーパーコピー代引き
Custom Packaging Bag
スーパーコピーバッグ
Desk-Free Luminaires
24 Birthday Candles
スーパーコピーバッグ