The Difficult Airway Algorithm?
Do we really need a difficult airway algorithm in the emergency department? I propose to you, that we approach every critically ill patient in the emergency department as a potentially difficult intubation. There is not one group that we can be more relaxed with and another that we apply a ‘difficult’ approach to. All are emergent intubations and all have the potential be difficult. Some patients will be anatomically difficult, some physiologically difficult and many will be both. What makes it challenging, is the small number of intubations we actually perform.
How many patients did you intubate last week? Some may have done an intubation, but the overwhelming majority would not have. This creates a potential problem. There might have been a time when we performed more intubations, however with Non-Invasive Ventilation (NIV) and high flow nasal cannula (HFNC) and perhaps the recent pandemic changing our thinking a little, we may not be intubating as often.
On average, an emergency physician performs 10 intubations per year, with about 25% doing less than 5(1). Without continued practice, there is a potential for us to fail, when we do need to intubate. Practicing this skill set, not only trains our motor skills, but it cognitively trains us, to flick the switch when needed and be able to go into ‘intubation/airway mode’.
We need to be to be strategic about what we practice. Our protocols/algorithms should take into account, the fact that we infrequently perform this procedure and we should make our salvage devices, those we bring out in cases of failed airway, our go-to devices. The techniques we use should be carefully selected and limited in number. They should be simple and intuitive and make our goal of first pass success easier. First pass success is associated with better clinical outcomes(2).
It’s not about learning several techniques such as direct laryngoscopy (DL), video laryngoscopy (VL) and fibre optics etc. It’s about becoming very good at one or two techniques that we can use when needed. Why is this important? Mostly because of the time it takes to learn a new technique.
Ericsson(3) says that we need 10,000 hours of practice to become expert in ‘something’. To put that into perspective. If we spent 8 hours per week, it would take us 24 years to reach that level. However we know, that we can utilise rapid skill acquisition techniques, such as deconstructing skills into subskills and practicing those, to attain a good skill level in approximately 20 hours(4). How good that level is, may vary.
The critically ill patient we see in the emergency department, is very different to the elective patient having a procedure in the operating theatre. The elective patients are screened and can potentially be woken up, if the tube is too difficult. Anaesthetists deal with airway every day, in every patient. However, when confronted with an acute airway in a critically ill patient, their stress levels also increase. An assessment of how difficult an airway is, adds nothing practically, when the patient needs to be intubated emergently.
The characteristics of our critically unwell patients, that make them a potentially difficult intubation include:
- Body Habitus (obese patient)
- Little respiratory reserve and thus desaturating quickly
- Haemodynamically unstable, or having the potential to be
- Depressed level of consciousness
Difficult Airway Algorithms
Multiple guidelines are available to assist us in planning for the difficult intubation in the critically unwell patient(5).
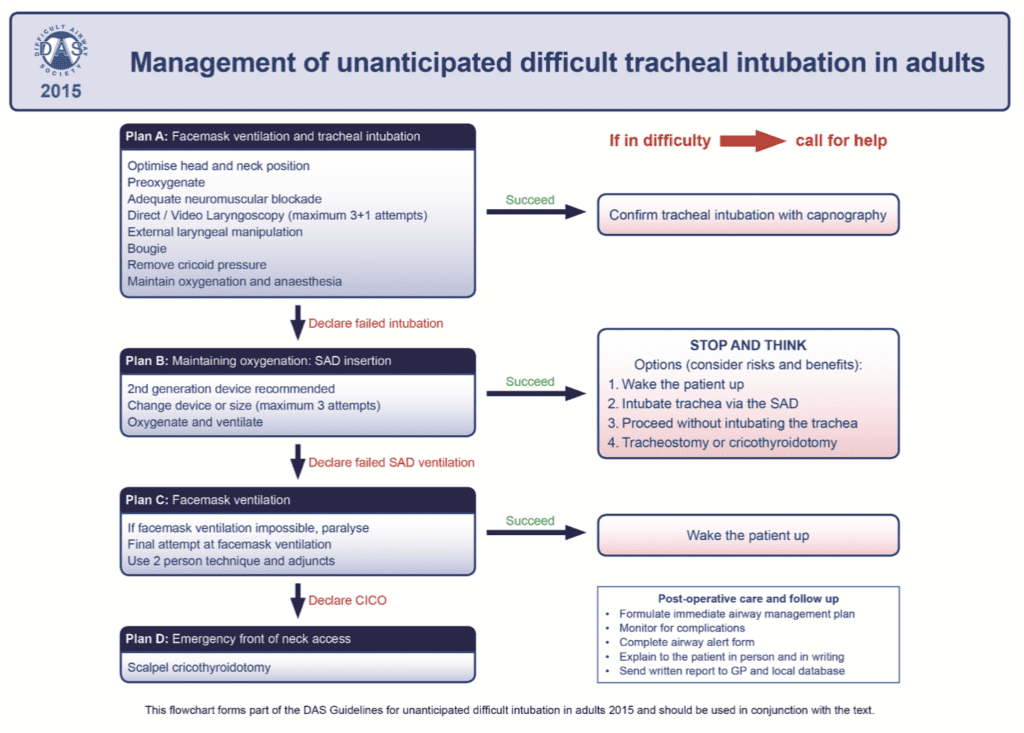
The algorithm shown in Figure 1 is from the Difficult Airway Society(6). It is a simple pathway that gives an overall approach, reinforcing that 3 failed attempts, is in fact a failed intubation and that we should then move to an alternative, such as supraglottic airway (SGA). The final step is always surgical airway. This pathway makes suggestions for, but leaves the final equipment choice to the operator.
The EMCrit team produced their own algorithm(7) shown in Figure 2 below, based on the Shocked Trauma Protocol. In this algorithm, the type of equipment and approach for each attempt at intubation is stipulated. This makes what to use and when, very clear. It is simple and straightforward.
It begins with a normal geometry VL with bougie and moves to a hyperangulated blade in the second attempt, then a senior performing the intubation on the third attempt. If still unable to intubate, a SGA is inserted. If failure to ventilate and oxygenate continues, a surgical airway is performed.
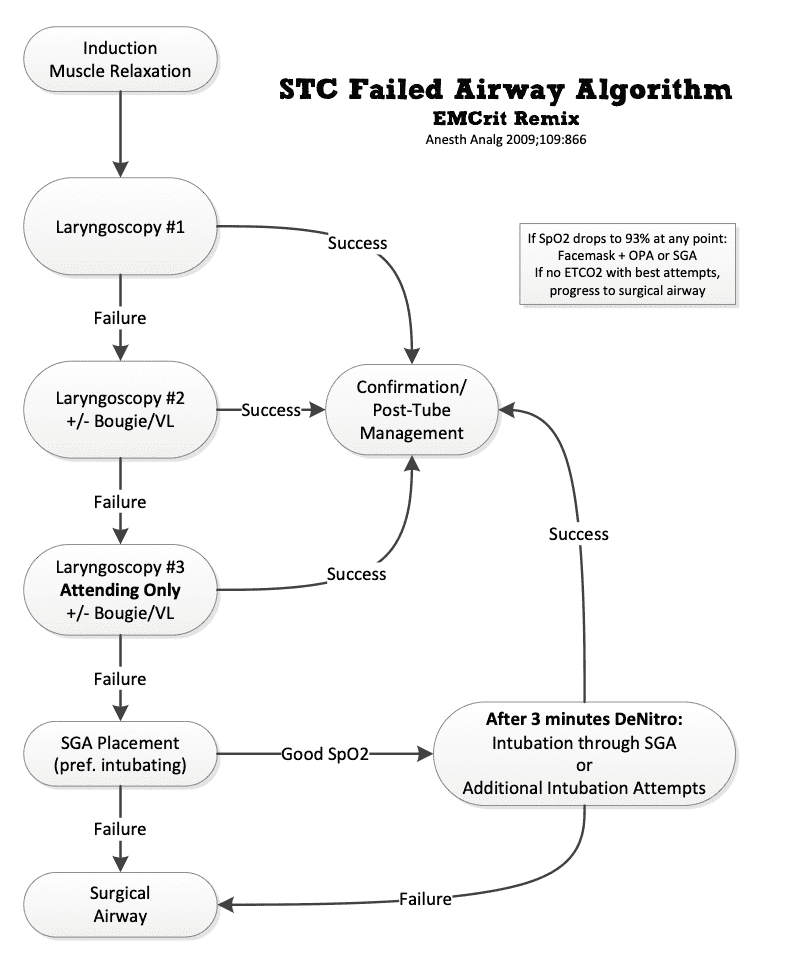
This STC failed airway algorithm provides an example of a protocol that can be used as is, or as a starting point for developing your own. It’s evidence based, but if you aren’t trained in hyperangulated blade VL, it can be an issue. The obvious solution is to get trained, otherwise you need to develop a different second attempt approach. I present a modified approach below.
Standard Geometry and Hyperangulated blades
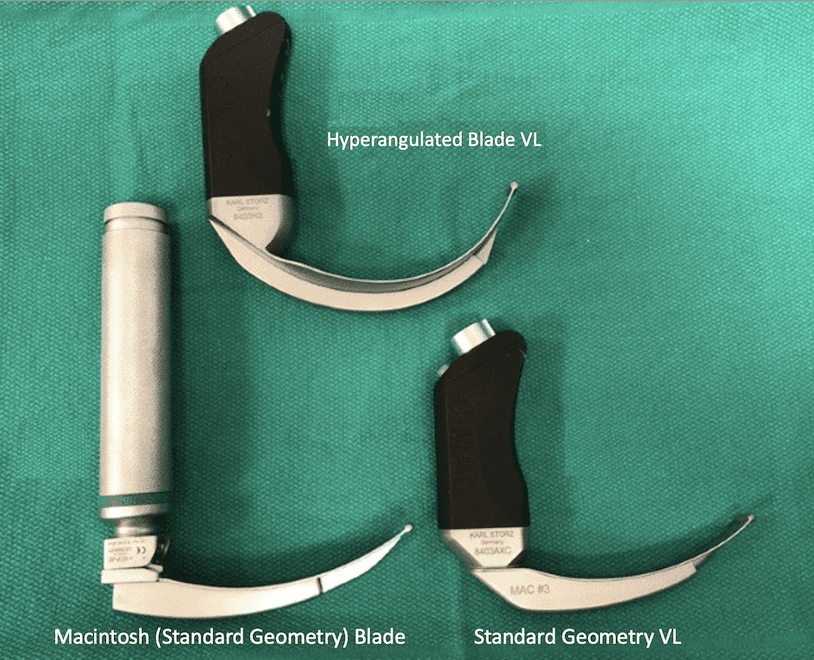
Direct Laryngoscopy uses a laryngoscope with a standard Mackintosh blade to displace the tongue and provide visualisation of the glottis. This is the standard approach that all of us learn.
Video Laryngoscopy uses two different blades, but with the ability to visualise the glottis on a screen. This can have benefits not only for the operator, but also for informing the rest of the team, as to the status of the intubation.
- The standard geometry blade
- This blade type is similar to the traditional DL mac blade and can be used in the same way. It can provide direct visualisation of the glottis.
- It also has the provision of being used as a video assist device.
- It can be used with a bougie or stylet
- The hyper-angulated blade
- This blade does not allow direct visualisation of the glottis. It is a pure video approach
- A bougie cannot be used with hyperangulated blade. A stylet is used.
How does DL compare to VL with a standard geometry blade?
Sakles (9) in a retrospective, observational single center study compared the Glidescope VL to DL. There were 823 patients with 62% in the DL group and 38% in the VL group. VL had a higher first pass success rate (75%) and less oesophageal intubations, even though overall the two devices were equivalent in terms of success rates.
Does standard geometry or hyperangulated VL give better first pass success rates?
Some institutions use a hyperangulated blade for their first attempt. Although I’m not sure where you go to on subsequent attempts, apart from changing the operator.
Driver looked at first attempt intubation success and compared standard geometry to hyperangulated VL (10). This was a prospective, multicenter, registry study of 11,927 intubations. It was not randomised. The two groups were not equal as the hyperangulated group had more patients with trauma, obesity and those with an initial impression of a difficult airway.
The results showed a 91.9% success rate with a standard geometry blade vs 89.2% for a hyperangulated. However, when results were adjusted for confounding, there was no difference.
The Bougie is an important part of intubation. Is it better than a stylet?
There are two camps: one pro-bougie and one pro-stylet. Is one really better than the other? I believe you should use what you are comfortable and experienced with. The bougie can be used with a standard geometry blade, but not with a hyperangulated blade. What does the evidence show?
Driver et al in 2018 (11) performed a randomised trial in a single center comparing bougie(n=381) to endotracheal tube and stylet (n=376).
The Primary outcome was first pass intubation success rate in patients with characteristics of a difficult airway, which included obesity, body fluids obstructing view, large tongue, facial trauma and others.
The primary outcome results of first pass success favoured the bougie ie 96% vs 82%. However this was a single center study, where a bougie was routines used for intubation, so we would expect that the operators would be more experienced in its use.
Driver repeated the study in 2021 (12) as a multicenter pragmatic randomised trial in 7 emergency departments and 8 ICUs in the US.
60% of intubations were performed by residents(registrars), 34% by fellows and 2% by attendings. VL was used 75% of the time.
First attempt success showed no statistically significant difference between bougie and entrotracheal tube with stylet: 80.4% vs 83% (p=0.27)
My take is use what you are most familiar and comfortable with.
The Difficult Airway Algorithm
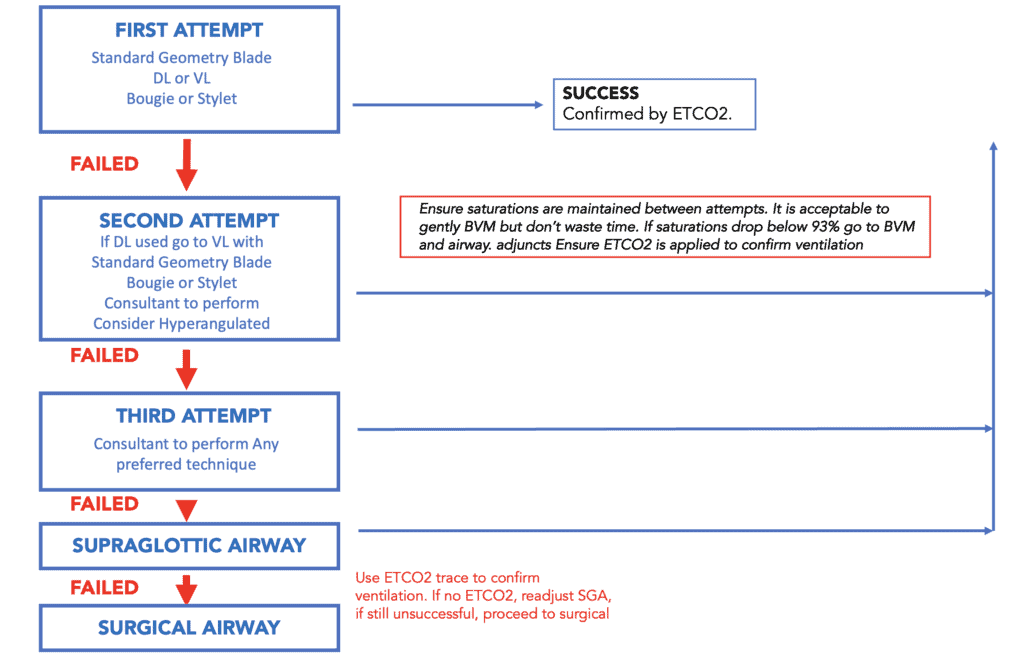
The following algorithm is based on the Difficult Airway Society and EMCrit algorithms, but with a few tweaks, that may decrease the need for learning new techniques as well as bringing in the senior to perform the intubation earlier.
The definition of a failed airway is 3 failed attempts at intubation. Following this, we would progress to a SGA, and if this failed, a surgical airway.
Following a failed attempt, we can bag valve mask (BVM) the patient, with or without a PEEP valve (I recommend using a PEEP valve), with ETCO2 attached (to ensure that we are ventilating). The endotracheal tube placement is confirmed by ETCO2 trace in all cases.
If the saturations drop at any point to below 93%, BVM with airway adjuncts can be used. Alternatively a SGA can be inserted and intubation can even be attempted through the SGA (depending on what type of SGA is used), following an improvement of the oxygen saturation.
The First Attempt
Use a standard geometry blade and either DL or VL with a bougie or stylet. If you are very confident with DL, then simply use it. However the option here, especially if you have flagged that this is a difficult situation, is to start with standard geometry blade and VL. A bougie or stylet can be used.
The Second Attempt
If the first attempt does not succeed, ask why?
If it was an issue with patient positioning especially in the obese patient, or excessive secretions? If so, we can correct those issues and can re-use the technique. If the glottis was not visualised on first attempt and DL was used, you can change blade sizes, however I would use VL with a standard geometry blade at that point. I would always have the consultant perform the second attempt. Sometimes a change in operator with the same technique can make all the difference.
The question that arises is, what to do for the second attempt, when VL with standard geometry was used in the first attempt. This is where training with a hyperangulated blade can be beneficial. It is certainly not the time to be using it for the first time. The technique needed for the hyperangulated blade differs markedly for that of a standard geometry blade with DL or VL. With this blade we move to pure video, no direct view. My approach is to change operator to a consultant at this time.
The Third Attempt
The third attempt must be performed by the most senior person available. They can use any technique they are comfortable with and they believe is appropriate for the situation.
When the Third Attempt Fails
If this fails, proceed to a SGA and ventilate the patient. Ensure that ventilation is effective, by using an ETCO2 trace. If ventilation is poor, adjusting the position of the SGA may significantly improve this.
If ventilation is possible with a SGA, then we can leave as is and wait for help, or we can attempt to intubate through the SGA. Alternatively once the patient’s saturations have increased, we can re-attempt intubation.
If unable to ventilate with a SGA, then proceed to surgical airway.
Regardless of which approach you use, have an algorithm, adapt it and train your whole team in it, so everyone is prepared for what is coming next.
References
- Carlson J N. Procedural Experience With Intubation: Results From a National Emergency Medicine Group. Ann Emeg Med 2019;74(6):786-794.
- Bernhard M et al. The first shot is often the Best Shot: First Pass Intubation Success in Emergency Airway Management. Anaeth and Analg 2015;121(5):pp1389-1393
- Ericsson K et al. The Cambridge Handbook of Expertise and Expert Performance. Cambridge University Press 2006.
- Kaufman J. The first 20 hours: How to learn anything fast. Penguin Books 2014.
- Higgs et al. Guidelines for the Management of Tracheal Intubation in Critically Ill Adults. BJA 2018;120(2):323-325
- C. Frerk, et al. Difficult Airway Society Intubation guidelines working group BJA, 2015; 115 (6): 827–848
- Scott Weingart, MD FCCM. Shock Trauma Center (STC) Failed Airway Algorithm. EMCrit Blog. Published on January 26, 2013
- Downey A et al.A systematic review of meta-analyses comparing direct laryngoscopy with video laryngoscopy. CJA. 2021; 68:706-714.
- Sakles JC et al. Tracheal Intubation in the Emergency Department: A comparison of Glidescope video laryngoscope to direct laryngoscopy in 822 intubations. J Emer Med. 2012;42(4): 400-405.
- Driver BE et al. Comparing Emergency Department First-Attempt Intubation Success with Standard-geometry and hyperangulated video laryngoscopes. Ann Emerg Med. 2020 Sept;76(3):332-338.
- Driver BE et al. Effect of Use of a Bougie vs Endotracheal Tube and Stylet on First-Attempt Intubation success among patients with difficult airways undergoing emergency intubation: A Randomised Clinical Trial. JAMA 2018;319(21) 2179-2186
- Driver BE et al. Effect of Use of a Bougie vs Endotracheal Tube and Stylet on First-Attempt Intubation success among patients with difficult airways undergoing emergency intubation: A Randomised Clinical Trial. JAMA 2021;326(24):2488-2497